Get Certified Today!
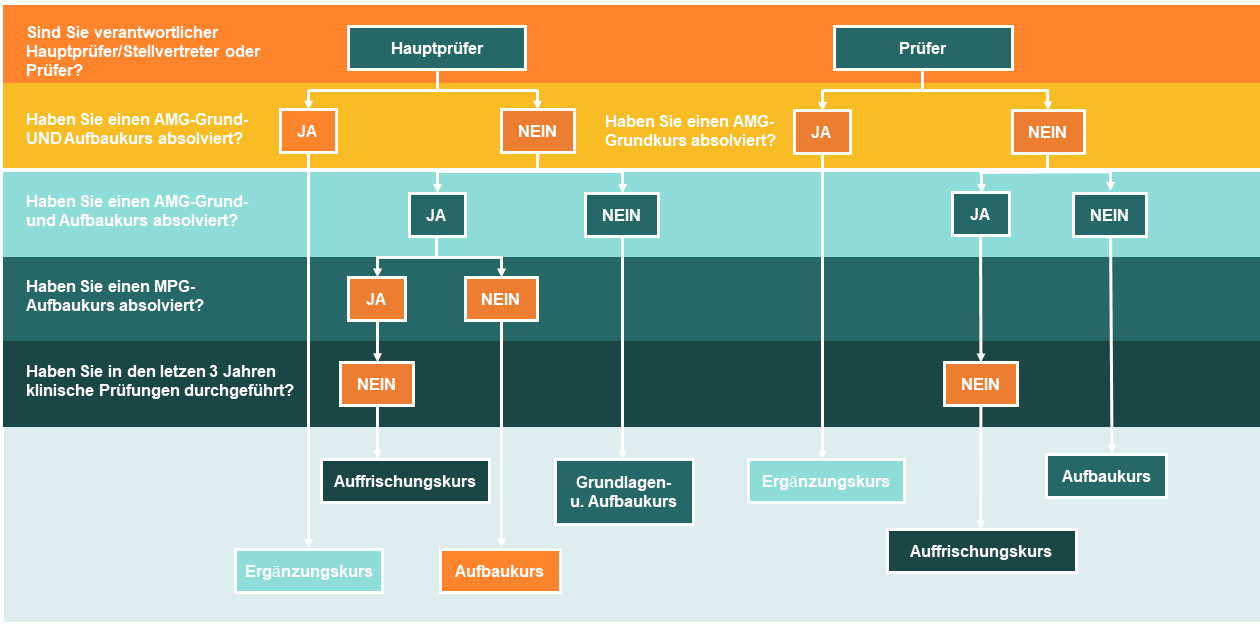
Do you need an MPG certificate to carry out clinical trials with medical devices? Would you like to deepen your knowledge? This Mastertrial MPG/GCP course is all online and is recognized by the Berlin Medical Association (CME). This Basic Course offers the following accredited training course for principal examiners, examiners and members of the examination group.
Download the MPG courses brochure








Reviews
There are no reviews yet.